APC RESISTANCE UNIVERSITY
Introduction | Biochemistry | Clinical Aspects | Key Reports | PubMed Abstracts | References | Assay Methods | APC Resistance Phenotyping | Factor V Mutation Screening | FAQ
INTRODUCTION
APC Resistance, a Common Risk Factor for Thrombosis
Activated protein C (APC) is a key anticoagulant enzyme needed for the proper down-regulation of blood coagulation. A poor anticoagulant response to APC, denoted APC resistance, is a recently described blood defect found to be a major risk factor for venous thromboembolism in Western societies. At least 90% of cases with the APC resistance phenotype can be explained by a point mutation in the coagulation factor V gene, changing arginine 506 in the factor V molecule to glutamine. After activation, mutated factor V (denoted FV:Q506 or Factor V Leiden) is partially resistant to inactivation by APC, which allows for longer duration of thrombin generation and may lead to a hypercoagulable state. APC resistance due to the presence of the FV:Q506 allele is inherited as an autosomal dominant trait and has a prevalence of 2-13% in the general population. Frequencies of APC resistance among patients with venous thrombosis, depending on the selection criteria, range from 20-60%. The high prevalence of APC resistance, combined with the availability of simple blood tests by which it is detected, raises the question of whether general screening for APC resistance should be performed in conjunction with circumstantial risk factors for thrombosis such as surgery, pregnancy and oral contraceptives.
Venous thromboembolism
The formation of an obstructive mass of clotted blood in the venous part of the circulatory system is known as venous thrombosis. The mass itself is called a thrombus and is composed of platelets, blood cells and fibrin. A thrombus which breaks loose and is carried away with the bloodstream is called an embolus. When caught in the blood vessels of the lung it may develop into pulmonary embolism, the most feared complication of venous thrombosis.1
Major health problem in Western countries
Venous thromboembolism is a major health problem in Western societies, constituting the third most common cardiovascular disease after acute ischemic heart disease and stroke.2 The incidence has increased steadily in recent centuries, perhaps due to longer life-spans and the adoption of more sedentary habits. In the USA, venous thromboembolism accounts for more than 250,000 hospitalizations a year, corresponding to an incidence of about one per 1,000 individuals. The annual death rate due to pulmonary embolism is estimated to be 50,000.2
Thrombogenic risk factors
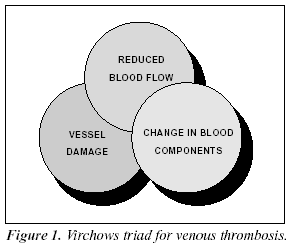
The three primary risk factors for venous thrombosis, as described by Virchow more than a century ago, are related to a reduced blood flow, vessel wall damage, and a change in blood components (Figure 1). Any one of these risk factors potentiates the other and may create a condition known as a hypercoagulable state. In principle, this condition can be viewed as a biochemical imbalance in the blood that favors blood coagulation and the formation of a blood clot.1,3
Risk factors for hypercoagulability and venous thromboembolism can be either acquired or inherited. Acquired risk factors such as surgery, pregnancy, oral contraceptives or immobilization are found in a large portion of patients. In addition, many patients also have a genetically determined tendency to thrombosis, as indicated by the fact that up to 40% of patients referred to a specialist laboratory have a family history of thrombosis.4-6 Until recently, three gene disorders had been identified associated with a clear increase in the risk for venous thromboembolism. These hereditary disorders were usually found in only a few percent of thrombosis-prone families through a symptomatic patient with a deficiency of either antithrombin, protein C or protein S.7 Thus, in vast majority of thrombotic patients no genetic risk factor could be identified.
Novel defect in the protein C anticoagulant pathway
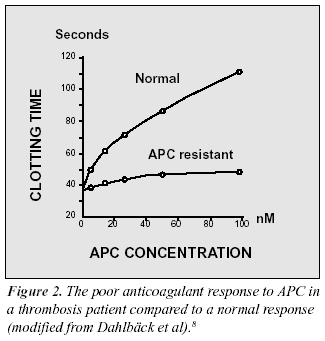
The diagnostic situation for familial thrombosis improved dramatically in 1993 with the discovery of a novel defect in the protein C anticoagulant pathway.8 Based on the hypothesis that a poor anticoagulant response to activated protein C (APC) might predispose to thrombosis, a Swedish research-team led by Björn Dahlbäck, investigated the activity of exogenously added APC in an APTT-based assay. In a normal response, the addition of APC to plasma induces a prolonged clotting time. This occurs because APC cleaves and inactivates two critical coagulation proteins, factors Va and VIIIa, resulting in a reduced rate of thrombin generation. However, when the assay was run on plasma from a middle-aged man suffering from recurrent episodes of venous thrombosis, the result showed a much shorter prolongation of the clotting time than expected (Figure 2). Several of the man’s relatives demonstrated the same phenotype, characterized by a poor anticoagulant response to APC. Family studies suggested that this blood disorder, denoted APC resistance, was inherited as an autosomal dominant trait.8,11 Subsequent investigations found that APC resistance was present in up to 60% of cases with a family history of venous thrombosis and that it was highly prevalent in the general population (1-7%).9-12 These results proved APC resistance to be the most common inherited defect associated with thrombosis, being larger than the sum of all other previously identified genetic risk factors.
Mutation in the factor V gene explains APC resistance
The search for the molecular mechanism of APC resistance led to the isolation of a protein from normal plasma, which was able to correct APC resistant plasma in a dose dependent manner. The protein was identified as factor V, suggesting that APC resistance was caused by a genetic defect in the factor V gene.37 Other studies reached the same conclusion and a point mutation that predicts the replacement of arginine at position 506 in the factor V molecule with glutamine was soon identified.13-15 At least 90% of APC resistant cases are explained by this mutation, denoted FV:Q506 or Factor V Leiden.16
Diagnostic breakthrough in thrombophilia
The discovery of APC resistance and the identification of the FV:Q506 mutation as its main cause means that a genetic explanation can be found almost as often as nongenetic risk factors in patients with venous thrombosis. A consequence of this diagnostic breakthrough has been a conceptual change in how thrombotic disease is viewed. In this monograph we attempt to summarize the knowledge gained during the last three years about APC resistance and to describe the major tests for its phenotype and genotype. It is our hope that the use of these tests will help establish guidelines for therapy and prophylaxis and lead to reduced morbidity and mortality in thrombosis-prone patients.
ORDER: 1-800-524-52224 | SUPPORT: 1-800-447-3846 | ABOUT US | CONTACT US | HOME
